Introduction to DEMO 2
Photochemistry is an important area of chemistry dealing with topics related to chemical reactions occurring under the irradiation of ultraviolet (UV), visible (Vis), or infrared (IR) radiation. The photochemistry includes topics focused on heterogeneous photocatalysis and photoelectrochemistry. Photocatalytic processes are used, among others, to remove pollutants from the liquid and gas phases, sterilize, produce self-cleaning coatings, or produce gases like hydrogen or oxygen.
In photoelectrocatalysis, photoelectrodes are utilized as working electrodes. They consist of a semiconductor layer and a current collector (usually transparent). Under the illumination of the semiconductor, a quasiparticle called an exciton is generated. The semiconductor intrinsically does not have charge carriers, but thanks to its properties and the appropriate design of the photoelectrode, the exciton is split into a hole and an electron. Depending on the semiconductor material and electrolyte composition, two types of photoelectrodes are distinguished: photocathode and photoanode. A photoanode is an electrode that utilizes holes generated from exciton splitting to drive oxidation reactions (e.g., H2O to O2). The complementary reduction process occurs at the cathode, driven by electrons transported from the photoanode.
In the UPTURN project, we work on the concept of a demonstrator called “DEMO 2” based on a TiO2 photoanode. A potential application is removing organic pollutants from aqueous solutions. Under the illumination of light of a specific wavelength, the titanium oxide photoanode generates reactive oxygen molecules, such as hydroxyl radicals, which are strong oxidants beneficial for the decomposition processes of organic compounds. Due to its short lifetime, it cannot be accumulated, so to use its properties, it must be produced in situ.
Initially, research demonstration work was carried out at a macroscopic scale. TiO2 anode was fabricated at the FTO glass substrate using titanium dioxide precursor, which is heated to burn off the polymer binder. The main advantage of this approach is a high specific surface area. The demonstration system consists of a Spectro-EFC 1.75 mL – Spectro-Electrochemical Flow Cell, an LED light source, a potentiostat, and a UV-Vis spectrophotometer. Methylene blue dye solution in 0.1M H2SO4 was applied as the electrolyte as a model reaction of removal of organic pollutants.
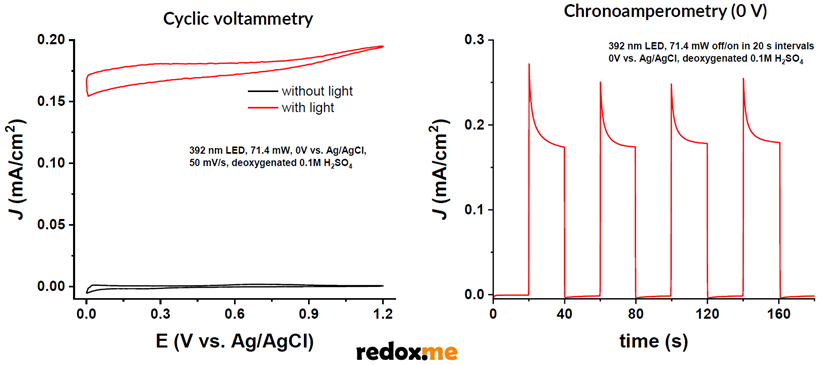
The photoelectrocatalytic effect was observed as the change in current measured at a given potential with and without illumination. CV scan recorded at the TiO2 anode electrode without illumination results in the current close to zero. Current recorded at the analogous conditions with light exposure exhibits a considerable increase (called photocurrent). In a chronoamperometric signal, if the TiO2 anode is kept at a constant voltage (e.g. 0 V), it can be observed that current only flows when the light source is turned on (see illustration below).
One of the aims of the UPTURN project is to develop microcells for the analysis of photoelectrochemical processes in flow. Currently, research work related to the miniaturization of the measurement system is being carried out.
To summarize, photoelectrocatalysis is an efficient method of converting light energy into chemical energy. This phenomenon can be used in many different fields, which provides a wide range of prospects for scientists carrying out work in the field of basic and industrial research.
Related products:
Spectro-EFC 1.75 mL – Spectro-Electrochemical Flow Cell



